Substantial progress on the afami-cel BLA including FDA agreement on plan for confirmatory evidence and favorable feedback on commercial T-cell potency assay - submission now targeted for Q4 2023
Transition of lete-cel from GSK and data from the completed pivotal trial in synovial sarcoma and MRCLS expected in late 2023
Initiated Phase 2 SURPASS-3 trial in ovarian cancer, which has the potential to be registrational and is supported by RMAT designation
Completed strategic combination with TCR2 to form a pre-eminent cell therapy company adding pipeline, technologies, approximately 40 people and $84.6m in Total Liquidity1 at closing
Company confirms pipeline data readouts over the next 18 months
Cash runway into early 2026
Webcast to be held today, August 9, 2023, at 8:00 a.m. EDT (1:00 p.m. BST)
Philadelphia, Pennsylvania and Oxford, United Kingdom--(Newsfile Corp. - August 9, 2023) - Adaptimmune Therapeutics plc (NASDAQ: ADAP), a leader in cell therapy to treat cancer, today reported financial results for the second quarter ended June 30, 2023 and provided a business update.
Adrian Rawcliffe, Adaptimmune's Chief Executive Officer: "The BLA submission process for afami-cel is going well with alignment with the Agency on key de-risking items. We have completed most of the wet work and we are now focused on writing and publishing the remaining sections to complete Part 3 of this submission for approval of the first engineered T-cell therapy for a solid tumor indication. We have completed the combination with TCR2 and added pipeline assets, technologies, and increased our total liquidity by approximately $85m. The transition of lete-cel back from GSK is also progressing well. We have set ourselves up to make data-driven portfolio decisions to bring medicines to market that we have high conviction can make a real difference for people with cancer."
Pipeline update and overview of near- and mid-term catalysts to make rigorous data-driven investment decisions
Adaptimmune's lead clinical franchises utilize engineered T-cell therapies targeting MAGE-A4, NY-ESO (in process of transitioning from GSK), and mesothelin, which are expressed on a broad range of solid tumors. Use of these cell therapies is supported by compelling clinical data including results in late-stage synovial sarcoma which will form the basis of the Company's first BLA submission. The Company has an enhanced "next-gen toolbox" and preclinical pipeline including PRAME and CD70 programs.
The following figure provides an overview of pipeline data catalysts that will be used to make data-driven investment decisions. To view an enhanced version of this graphic, please visit: https://images.newsfilecorp.com/files/8845/168338_adptimage.jpg. Note, this figure was previously provided in the Adaptimmune corporate deck and a press release issued on June 1st to announce completion of the strategic combination with TCR2.
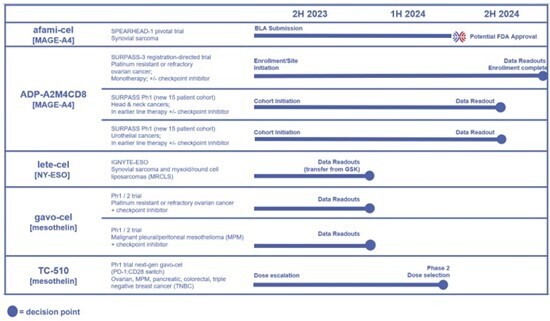
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/8845/176502_figure1.jpg
Afami-cel targeting MAGE-A4
- Parts 1 and 2 of the afami-cel BLA submission have been completed and the final module (Part 3) is in progress. Milestone achievements are outlined in the BLA update section below.
Lete-cel targeting NY-ESO (in process of transitioning from GSK): could offer another potential commercial treatment for people with sarcoma
- Data from the completed pivotal clinical trial with lete-cel (IGNYTE-ESO) for the treatment of synovial sarcoma or myxoid/ round cell liposarcoma (MRCLS) is expected in late 2023. The transition of lete-cel to Adaptimmune from GSK is ongoing and terms were agreed during Q2 2023. The Company will determine next steps for lete-cel, including potential for a BLA, in early 2024.
ADP-A2M4CD8: next-generation therapy targeting MAGE-A4
- Initiated the Phase 2 SURPASS-3 trial in combination with nivolumab for platinum resistant ovarian cancer. This trial has the potential to become registrational. ADP-A2M4CD8 has been granted FDA RMAT designation for the treatment of people with platinum resistant ovarian cancer.
- Initiated additional cohorts in the Phase 1 SURPASS trial in the earlier line treatment setting for head & neck and urothelial cancers in combination with checkpoint inhibitors. Data readouts anticipated in 2H 2024.
- An update on the Phase 1 SURPASS trial (ADP-A2M4CD8 alone or combined with nivolumab) will be featured as an oral presentation at the European Society of Medical Oncology (ESMO) 2023 Congress, Monday, October 23, 11:08-11:18 CET.
Gavo-cel targeting mesothelin
- The Phase 2 portion of the trial to assess gavo-cel in people with ovarian cancer in combination with nivolumab is ongoing. All patients to be included in an interim data review have been enrolled. All enrolled patients will be treated and continue in the trial as planned. Additional patients will not be enrolled until the data review is complete.
- Note, the Phase 1 portion of the trial in ovarian cancer, non-small cell lung cancer (NSCLC), malignant pleural/peritoneal mesothelioma (MPM) or cholangiocarcinoma has been completed. Initial data from the Phase 1 portion of the trial was reported in September 2022. Focus was subsequently narrowed to ovarian cancer.
- More mature clinical and translational Phase 1 and 2 data will be evaluated later this year to determine next steps with gavo-cel.
TC-510: next-generation therapy targeting mesothelin
- The Phase 1/2 clinical trial for TC-510 for people with MPM, ovarian cancer, pancreatic cancer, colorectal cancer, or triple negative breast cancer is ongoing in the dose-escalation phase. Initial data readouts are anticipated this year to enable further directional decisions for TC-510.
Preclinical programs targeting PRAME and CD70 (not shown in figure)
- Adaptimmune plans to be IND-ready in late 2023 with a PRAME targeted TCR T-cell therapy. PRAME is a clinically validated target which is highly expressed across a broad range of solid tumors including breast, kidney, endometrial, ovarian, gastroesophageal, non-small cell lung, and head & neck cancers as well as melanoma. A clinical trial is expected to initiate in 2024 (indications remain to be determined).
- An IND for CD70 is planned for 2024. CD70 is expressed in hematological malignancies including acute myeloid leukemia (AML), lymphoma, and solid tumors including renal cell carcinoma.
Afami-cel: Adaptimmune plans to complete BLA submission in Q4 2023
In late 2022, Adaptimmune completed submission of the preclinical module (Part 1) of the BLA. In Q1 2023, the Company completed submission of the clinical module (Part 2). The Company is currently in the process of completing the final CMC module (Part 3).
Recently, several critical milestones have been completed:
- Demonstrated comparability of afami-cel drug batches manufactured with clinical trial supply vector and commercial supply vector.
- In collaboration with our companion diagnostic (CDx) partner, completed submission to FDA of the Premarket application (PMA) for the MAGE-A4 CDx assay.
- Agreed confirmatory evidence plan with FDA: Cohort 2 of the SPEARHEAD-1 trial will act as confirmatory evidence for full approval, enrollment in Cohort 2 has completed.
- Completed method validation for lot release assays (including potency assays).
- Completed vector process performance qualification (PPQ).
- Initiated T-cell process performance qualification (PPQ).
- Secured favorable FDA feedback on the commercial T-cell potency assay including agreement on the proposed potency dataset for inclusion in the submission. The FDA has not requested any new or additional information on T-cell potency to precede the BLA submission.
This BLA is supported by data from Cohort 1 of the pivotal trial SPEARHEAD-1, which met its primary endpoint for efficacy. The Company has Regenerative Medicine Advanced Therapy (RMAT) designation from the FDA for afami-cel for the treatment of synovial sarcoma.
Afami-cel data presentation at ASCO 2023
Data from SPEARHEAD-1 was presented in a poster at the American Society of Clinical Oncology (ASCO) Annual Meeting, which is available HERE and summarized below.
- Approximately 39% of patients had clinical responses after a single dose of afami-cel in Cohort 1 of the pivotal SPEARHEAD-1 trial and the median duration of response was ~12 months (CTOS 2022).
- Median overall survival (mOS) was ~17 months.
- Patients with a RECIST response have a 12-month OS probability of 90% and 24-month OS probability of 70%, and the mOS for responders has not yet been reached.
- Historical outcomes are poor for advanced synovial sarcoma with a mOS of <12 months in the second line and beyond treatment setting.
Corporate news
- In June, Adaptimmune announced completion of the strategic combination with TCR2 to form a pre-eminent cell therapy company . At completion of the combination an additional $84.6m of Total Liquidity1 was added to the group's combined liquidity and 39 additional employees. Integration efforts, including pipeline prioritization and investment decisions, are ongoing.
- Adaptimmune and GSK agreed terms for transfer of PRAME and NY-ESO ("lete-cel") target programs back to Adaptimmune in Q2. Adaptimmune will receive ~$37 million from GSK in relation to the transition of the ongoing lete-cel clinical trials anticipated in Q4.
Financial Results for the three and six months ended June 30, 2023
- Cash / liquidity position: As of June 30, 2023, Adaptimmune had cash and cash equivalents of $77.0 million and Total Liquidity1 of $204.7 million, compared to $108.0 million and $204.6 million, respectively, as of December 31, 2022.
- Revenue: Revenue for the three and six months ended June 30, 2023, was $5.1 million and $52.7 million, respectively, compared to $5.5 million and $9.1 million for the same periods in 2022. Revenue has increased in the six months to June 30, 2023, compared to the same period in 2022 primarily due to the termination of the Astellas collaboration, resulting in the remaining deferred income for the collaboration being released as revenue in March 2023.
- Research and development (R&D) expenses: R&D expenses for the three and six months ended June 30, 2023, were $30.0 million and $55.5 million, respectively, compared to $34.7 million and $71.5 million for the same periods in 2022. R&D expenses decreased due to a decrease in the average number of employees engaged in research and development, decreases in subcontracted expenditures, a decrease in share-based compensation expenses, a decrease in in-process research and development costs and a decrease in offsetting reimbursements receivable for research and development tax and expenditure credits.
- General and administrative (G&A) expenses: G&A expenses for the three and six months ended June 30, 2023, were $20.1 million and $40.5 million, respectively, compared to $14.6 million and $31.4 million for the same periods in 2022. G&A expenses increased due to restructuring and charges recognised in the first quarter of 2023, an increase in other corporate costs due to an increase in accounting, legal and professional fees incurred in relation to the TCR2 Therapeutics, Inc merger agreement and severance and other related costs for former TCR2 Therapeutics leadership, offset by a decrease in share-based compensation expenses.
- Gain on bargain purchase: a $22.2 million gain on bargain purchase was recognised in the three and six months ended June 30, 2023, from the strategic combination with TCR2 Therapeutics, Inc.
- Net loss: Net loss attributable to holders of the Company's ordinary shares for the three and six months ended June 30, 2023, was $21.4 million and $20.4 million, respectively ($(0.02) and $(0.02) per ordinary share), compared to $44.5 million and $94.8 million, respectively ($(0.05) and $(0.10) per ordinary share), for the same periods in 2022.
Financial Guidance
The Company believes that its existing cash, cash equivalents and marketable securities, together with the additional payments under the Strategic Collaboration and License Agreement with Genentech and payments under the Termination and Transfer Agreement with GSK, will fund the Company's current operations into early 2026, as further detailed in the Company's Quarterly Report on Form 10-Q for the second quarter ended June 30, 2023, to be filed with the Securities and Exchange Commission following this earnings release.
Webcast Information
The Company will host a live webcast to provide additional details at 8:00 a.m. EDT (1:00 p.m. BST) today, August 9, 2023. A live webcast of the conference call and replay can be accessed at https://www.gowebcasting.com/12658. Call in information is as follows: 1-800-806-54874 (US or Canada) or +1 (416)-340-2217 participant passcode 4991002# (International and additional options available HERE). Callers should dial in 5-10 minutes prior to the scheduled start time and simply ask to join the Adaptimmune call.
About Adaptimmune
Adaptimmune is a clinical-stage biopharmaceutical company focused on the development of novel cancer immunotherapy products for people with cancer. The Company's cell therapy products have shown clinical responses in multiple solid tumor indications. Our unique T-cell platforms enable us to identify cancer targets, find and develop cell therapy candidates active against those targets, and produce therapeutic candidates for administration to patients. Adaptimmune's cell therapy products include T-cells with genetically engineered T-cell receptors ("TCR T-cells"), TCR Fusion Construct T cells (TRuC-T cells), and HLA-independent TCRs ("HiTs").
Forward-Looking Statements
This release contains "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995 (PSLRA). These forward-looking statements involve certain risks and uncertainties. Such risks and uncertainties could cause our actual results to differ materially from those indicated by such forward-looking statements, and include, without limitation: the success, cost and timing of our product development activities and clinical trials and our ability to successfully advance our TCR therapeutic candidates through the regulatory and commercialization processes. For a further description of the risks and uncertainties that could cause our actual results to differ materially from those expressed in these forward-looking statements, as well as risks relating to our business in general, we refer you to our Annual Report on Form 10-K for the year ended December 31, 2022 filed with the Securities and Exchange Commission (SEC) on March 6, 2023, our Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, and other filings with the SEC. The forward-looking statements contained in this press release speak only as of the date the statements were made and we do not undertake any obligation to update such forward-looking statements to reflect subsequent events or circumstances.
Total Liquidity (a non-GAAP financial measure)
Total Liquidity (a non-GAAP financial measure) is the total of cash and cash equivalents and marketable securities (available-for-sale debt securities). Each of these components appears separately in the condensed consolidated balance sheet. The U.S. GAAP financial measure most directly comparable to Total Liquidity is cash and cash equivalents as reported in the condensed consolidated financial statements, which reconciles to Total Liquidity as follows (in thousands):
| June 30, | December 31, | |||||
| 2023 | 2022 | |||||
| Cash and cash equivalents | $ | 76,969 | $ | 108,033 | ||
| Marketable securities - available-for-sale debt securities | 127,738 | 96,572 | ||||
| Total Liquidity | $ | 204,707 | $ | 204,605 | ||
The Company believes that the presentation of Total Liquidity provides useful information to investors because management reviews Total Liquidity as part of its assessment of overall solvency and liquidity, financial flexibility, capital position and leverage.
Condensed Consolidated Statement of Operations
(unaudited, in thousands, except per share data)
| Three months ended | Six months ended | |||||||||||
| June 30, | June 30, | |||||||||||
| 2023 | 2022 | 2023 | 2022 | |||||||||
| Revenue | $ | 5,130 | $ | 5,538 | $ | 52,731 | $ | 9,113 | ||||
| Operating expenses | ||||||||||||
| Research and development | (29,965) | (34,740) | (55,513) | (71,492) | ||||||||
| General and administrative | (20,073) | (14,550) | (40,470) | (31,354) | ||||||||
| Total operating expenses | (50,038) | (49,290) | (95,983) | (102,846) | ||||||||
| Operating loss | (44,908) | (43,752) | (43,252) | (93,733) | ||||||||
| Interest income | 1,543 | 357 | 2,219 | 695 | ||||||||
| Gain on bargain purchase | 22,155 | - | 22,155 | - | ||||||||
| Other income (expense), net | 501 | (655) | (170) | (643) | ||||||||
| Loss before income tax expense | (20,709) | (44,050) | (19,048) | (93,681) | ||||||||
| Income tax expense | (680) | (470) | (1,305) | (1,104) | ||||||||
| Net loss attributable to ordinary shareholders | $ | (21,389) | $ | (44,520) | $ | (20,353) | $ | (94,785) | ||||
| | ||||||||||||
| Net loss per ordinary share | ||||||||||||
| Basic and diluted | $ | (0.02) | $ | (0.05) | $ | (0.02) | $ | (0.10) | ||||
| | ||||||||||||
| | ||||||||||||
| Weighted average shares outstanding: | ||||||||||||
| Basic and diluted | 1,108,166,960 | 962,794,072 | 1,050,071,434 | 951,474,546 | ||||||||
| | ||||||||||||
Condensed Consolidated Balance Sheets
(unaudited, in thousands, except share data)
| June 30, | December 31, | |||||
| 2023 | 2022 | |||||
| Assets | ||||||
| Current assets | ||||||
| Cash and cash equivalents | $ | 76,969 | $ | 108,033 | ||
| Marketable securities - available-for-sale debt securities | 127,738 | 96,572 | ||||
| Accounts receivable, net of allowance for expected credit losses of $0 and $0 | 2,970 | 7,435 | ||||
| Other current assets and prepaid expenses | 54,094 | 43,330 | ||||
| Total current assets | 261,771 | 255,370 | ||||
| | ||||||
| Restricted cash | 3,231 | 1,569 | ||||
| Operating lease right-of-use assets, net of accumulated amortization of $11,258 and $9,470 | 22,027 | 18,019 | ||||
| Property, plant and equipment, net of accumulated depreciation of $40,635 and $38,588 | 55,492 | 53,516 | ||||
| Intangible assets, net of accumulated amortization of $5,003 and $4,676 | 463 | 442 | ||||
| Total assets | $ | 342,984 | $ | 328,916 | ||
| | ||||||
| Liabilities and stockholders' equity | ||||||
| Current liabilities | ||||||
| Accounts payable | $ | 14,713 | $ | 4,753 | ||
| Operating lease liabilities, current | 4,752 | 2,728 | ||||
| Accrued expenses and other current liabilities | 25,242 | 31,215 | ||||
| Restructuring provision | - | 2,285 | ||||
| Deferred revenue, current | 31,418 | 23,520 | ||||
| Total current liabilities | 76,125 | 64,501 | ||||
| | ||||||
| Operating lease liabilities, non-current | 21,590 | 20,349 | ||||
| Deferred revenue, non-current | 117,257 | 160,892 | ||||
| Other liabilities, non-current | 1,361 | 1,296 | ||||
| Total liabilities | 216,333 | 247,038 | ||||
| | ||||||
| Stockholders' equity | ||||||
| Common stock - Ordinary shares par value £0.001, 1,702,760,280 authorized and 1,351,828,044 issued and outstanding (2022: 1,282,773,750 authorized and 987,109,890 issued and outstanding) | 1,851 | 1,399 | ||||
| Additional paid in capital | 1,057,547 | 990,656 | ||||
| Accumulated other comprehensive loss | (3,092) | (875) | ||||
| Accumulated deficit | (929,655) | (909,302) | ||||
| Total stockholders' equity | 126,651 | 81,878 | ||||
| | ||||||
| Total liabilities and stockholders' equity | $ | 342,984 | $ | 328,916 | ||
Condensed Consolidated Cash Flow Statement
(unaudited, in thousands)
| Six months ended | ||||||
| June 30, | ||||||
| 2023 | 2022 | |||||
| Cash flows from operating activities | ||||||
| Net loss | $ | (20,353) | $ | (94,785) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||
| Depreciation | 3,824 | 2,728 | ||||
| Amortization | 253 | 419 | ||||
| Gain on bargain purchase | (22,155) | - | ||||
| Share-based compensation expense | 5,513 | 10,631 | ||||
| Unrealized foreign exchange losses/(gains) | 377 | (108) | ||||
| (Accretion)/amortization on available-for-sale debt securities | (633) | 1,636 | ||||
| Other | 663 | 585 | ||||
| Changes in operating assets and liabilities: | ||||||
| Decrease/(increase) in receivables and other operating assets | 1,971 | (22,898) | ||||
| (Decrease)/increase in payables and other current liabilities | (8,801) | 12,898 | ||||
| Decrease in deferred revenue | (41,704) | (6,758) | ||||
| Net cash used in operating activities | (81,045) | (95,652) | ||||
| Cash flows from investing activities | ||||||
| Acquisition of property, plant and equipment | (3,565) | (16,074) | ||||
| Acquisition of intangible assets | (199) | - | ||||
| Acquired upon acquisition of TCR2 Therapeutics Inc. | 45,264 | - | ||||
| Maturity or redemption of marketable securities | 76,119 | 97,605 | ||||
| Investment in marketable securities | (67,121) | (42,197) | ||||
| Other | 537 | - | ||||
| Net cash provided by investing activities | 51,035 | 39,334 | ||||
| Cash flows from financing activities | ||||||
| Proceeds from issuance of common stock from offerings, net of commissions and issuance costs | 188 | 9,976 | ||||
| Proceeds from exercise of stock options | 22 | 36 | ||||
| Net cash provided by financing activities | 210 | 10,012 | ||||
| Effect of currency exchange rate changes on cash, cash equivalents and restricted cash | 398 | (5,836) | ||||
| Net decrease in cash, cash equivalents and restricted cash | (29,402) | (52,142) | ||||
| Cash, cash equivalents and restricted cash at start of period | 109,602 | 151,666 | ||||
| Cash, cash equivalents and restricted cash at end of period | $ | 80,200 | $ | 99,524 | ||
Adaptimmune Contact
Investor Relations
Juli P. Miller, Ph.D. - VP, Corporate Affairs and Investor Relations
T: +1 215 825 9310
M: +1 215 460 8920
[email protected]
Media Relations
Dana Lynch, Senior Director of Corporate Communications
M: +1 267 990 1217
[email protected]
1 Total liquidity is a non-GAAP financial measure, which is explained and reconciled to the most directly comparable financial measures prepared in accordance with GAAP below.

To view the source version of this press release, please visit https://www.newsfilecorp.com/release/176502

